Clinical utility of prognostic AMBLor® technology demonstrated in US patient case study
In recognition of Melanoma Awareness Month, AMLo Biosciences are sharing a patient case study demonstrating the clinical utility of AMBLor®, their technology that accurately stratifies early-stage low-risk melanomas. Using the biomarkers loricrin and AMBRA1, the simple laboratory assay* that can be performed on a patient’s initial skin sample identifies a subset of melanomas that are at low risk of progression.
Why is there a need to understand melanoma progression risk?
Skin cancers are on the rise and The American Cancer Society estimates around 100,640 new melanomas will be diagnosed in 2024[1]. Melanoma is the deadliest form of skin cancer and by 2040, it is estimated that melanoma will be the 2nd most common cancer in the USA[2].
The current diagnostic pathway for the clinical staging of melanomas uses information obtained from clinical and radiological assessment, including a tumor biopsy. The melanoma category is determined based on Breslow tumor thickness and the presence or absence of ulceration[3]. Further prognostic information is gained through additional procedures such as sentinel lymph node biopsy (SLNB), local tumor excision, and lymph node dissection[3], but meta-analysis data have shown that on average 12.5% of SLNBs can produce a false-negative result[4]. Currently available gene expression profiling (GEP) tests are not always considered to provide enough additional independent prognostic information[5].
The American Cancer Society estimates 8,290 people will die of melanoma in 2024[1], of which about 40% will have initially been diagnosed with Stage I or II melanoma[6]. It is therefore imperative to better stratify the risk of progression in early-stage melanoma patients in order to deliver effective treatment and monitoring on a patient-by-patient basis. AMBLor technology can be used to determine the pre-ulcerative status and it is independent of Breslow depth to accurately provide physicians with greater insight into a patient’s individual prognosis.
Supplying health care providers with additional data early in their patient’s melanoma journey can result in a more tailored, personalized approach to treatment and follow-up. Additionally, it may make a difference in patient survival years and quality of life, along with impacting utilization of downstream resources.
Read the full case study below to see how AMBLor can provide substantial benefit to clinical practice in melanoma management.
References
- American Cancer Society. About Melanoma Skin Cancer Factsheet. 2024. https://www.cancer.org/content/dam/CRC/PDF/Public/8823.00.pdf. Accessed May 2024
- Rahib L, Wehner MR, Matrisian LM, Nead KT. Estimated Projection of US Cancer Incidence and Death to 2040. JAMA Netw Open. 2021;4(4):e214708. doi: www.10.1001/jamanetworkopen.2021.4708
- Pathak S & Zito PM. Clinical Guidelines for the Staging, Diagnosis, and Management of Cutaneous Malignant Melanoma. [Updated 2023 Jun 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK572149/
- Valsecchi ME et al. Lymphatic mapping and sentinel lymph node biopsy in patients with melanoma: a meta-analysis. J Clin Oncol 2011;29(11):1479-1487. doi: www.10.1200/JCO.2010.33.1884
- Grossman D et al. Prognostic Gene Expression Profiling in Cutaneous Melanoma: Identifying the Knowledge Gaps and Assessing the Clinical Benefit. JAMA Dermatol 2020;156(9):1004-1011. doi: www.10.1001/jamadermatol.2020.1729
- Zhou C, et al. Primary Melanoma Characteristics of Metastatic Disease: A Nationwide Cancer Registry Study. Cancers (Basel). 2021;13(17):4431. doi: www.10.3390/cancers13174431
A Case Study on the Use of a Novel Prognostic Biomarker for Melanoma
AUTHORS
Dr. Maureen Basius, D.O.
Consultant Pathologist, AMLo Biosciences, US
Jennifer Goodman, M.Sc.
Medical Science Liaison, AMLo Biosciences, US
Introduction
This review is being written as a retrospective individual case study examining the use of the AMBLor® test, a commercially available prognostic test for early-stage melanoma. AMBLor looks for the presence or absence of two proteins, AMBRA1 and Loricrin. The test comprises a dual antibody IHC stain applied to sections from the diagnostic block of a non-ulcerated cutaneous melanoma that has been assigned an AJCC staging of IA–IIB. The assay is read by a trained histopathologist in conjunction with the H&E slide of the same tissue.
AMBRA1 and Loricrin are proteins normally expressed in the epidermis of the skin. They have important roles in epidermal differentiation and therefore maintenance of epidermal integrity. In normal skin and specifically the epidermis, AMBRA1 expression levels increase in parallel with keratinocyte differentiation from the basal layer to the stratum corneum, while Loricrin is expressed as a single line of staining in the stratum corneum containing terminally differentiated keratinocytes. The secretion of the signaling molecule, transforming growth factor-beta 2 (TGFß2) by higher risk AJCC Stage I or II melanomas, has been shown to cause downregulation of AMBRA1 and Loricrin as well as other proteins (e.g. the gap junctional protein, claudin-1). Loss of AMBRA1 and Loricrin results in loss of epidermal integrity, thereby facilitating tumor erosion of the epidermis and identifying a pre-ulcerative state.
Patient History
The patient is an 83-year-old Caucasian male with past medical history of Stage I colon cancer (resected 2003), low grade prostatic cancer (post seed implantation 2007), Chronic Myelogenous Leukemia, chronic phase (bone marrow aspirate and biopsy 2015), and paroxysmal atrial fibrillation (controlled).
The patient presented to his dermatologist in June 2022 with a chief complaint of two atypical melanocytic lesions located upon his back. The first lesion was located in the left lateral upper back and a second lesion was located on the superior thoracic spine region. During his initial visit, a shave biopsy was performed to remove both lesions.
The left lateral upper back malignant Melanoma was found to be extending to peripheral and deep margins, superficial spreading type, at least 0.8mm thickness, negative for surface ulceration with a pathologic stage at least pT1b. The second lesion superior thoracic spine malignant Melanoma in situ with a pathologic stage pTis.
In July 2022 the patient underwent complete wide excision of both lesions. The left lateral upper back malignant Melanoma resulted in NOS Breslow thickness: 14.5mm Anatomic/Clark Level V. Margins were free of melanoma and melanoma in situ. The superior thoracic spine Melanoma in situ resulted in NOS Anatomic/Clark level I. The patient subsequently underwent a left axillary sentinel lymph node excision resulting in one benign lymph node. Immunohistochemical staining on the left axillary sentinel lymph node for tyrosinase, HMB45 and MART-1 was negative for metastatic melanoma.
In July 2022, the patient’s oncologist had a detailed discussion with the patient and his family, explaining that based on all available testing, the patient’s prognosis was “excellent”, while also conveying a recurrence rate risk of at least 20%. The patient’s oncologist advised that the use of adjuvant checkpoint inhibitor therapy (given as pembrolizumab 200 mg meter squared q3 weeks times 12 months) would improve the chance of recurrence to 10%. After thoughtful consideration, the patient opted not to proceed with adjuvant therapy.
In January 2023, the patient presented to an urgent care facility with cough and shortness of breath. Subsequent chest X-ray revealed dense nodularity throughout the lungs bilaterally. Upon further radiologic scans and testing, the patient was found to have metastatic melanoma to the liver, lungs, and retroperitoneal lymph nodes accompanied by a malignant pleural effusion. The patient immediately began treatment with ipilimumab and nivolumab but was unresponsive to therapy. The patient’s condition rapidly deteriorated and the patient expired May 17th 2023.
AMBLor Testing and Guidelines
AMBLor looks for the presence or absence of two proteins, AMBRA1 and Loricrin, in the epidermis overlying a specific lesion. There are 4 possible outcomes of a test and two results:
Result 1: “low-risk”
- Outcome 1: AMBRA1 maintained / Loricrin maintained
- Outcome 2: AMBRA1 maintained / Loricrin lost
- Outcome 3: AMBRA1 lost / Loricrin maintained
Result 2: “at-risk”
- Outcome 4: AMBRA1 lost / Loricrin lost
A low-risk test score means that one or both of the AMBRA1 and Loricrin proteins were maintained (present) in the skin overlying the melanoma. Published data associates a low-risk test score with melanomas that have not progressed. These data show that a low-risk result is 97% (NPV) accurate with up to 12 years clinical follow up data.[4] In contrast the 8th edition AJCC guidelines suggest that Stage IA-IIB melanomas have between a 98–82% chance of being disease free after 10 years.
An at-risk test score means that both AMBRA1 and Loricrin proteins are at least focally lost (absent) from the epidermis overlying the melanoma. The test has a low positive predictive value (PPV) which means it does not identify melanomas at high risk of progression to metastatic disease. It is probable that the high false positive rate is due to successful surgical excision of an at-risk tumor, which means the patient was recurrence free at analysis. Patients assigned an at-risk score should therefore be managed according to the current guidelines.
For the patient in question, with family permission, AMBLor was performed postmortem upon recuts of the formalin fixed paraffin block of the left lateral upper back lesion. Both AMBRA1 and Loricrin were lost in this lesion, indicating that the patient’s left lateral upper back lesion was “AT-RISK”. The at-risk outcome is consistent with a level of risk of metastasis associated with current AJCC staging.
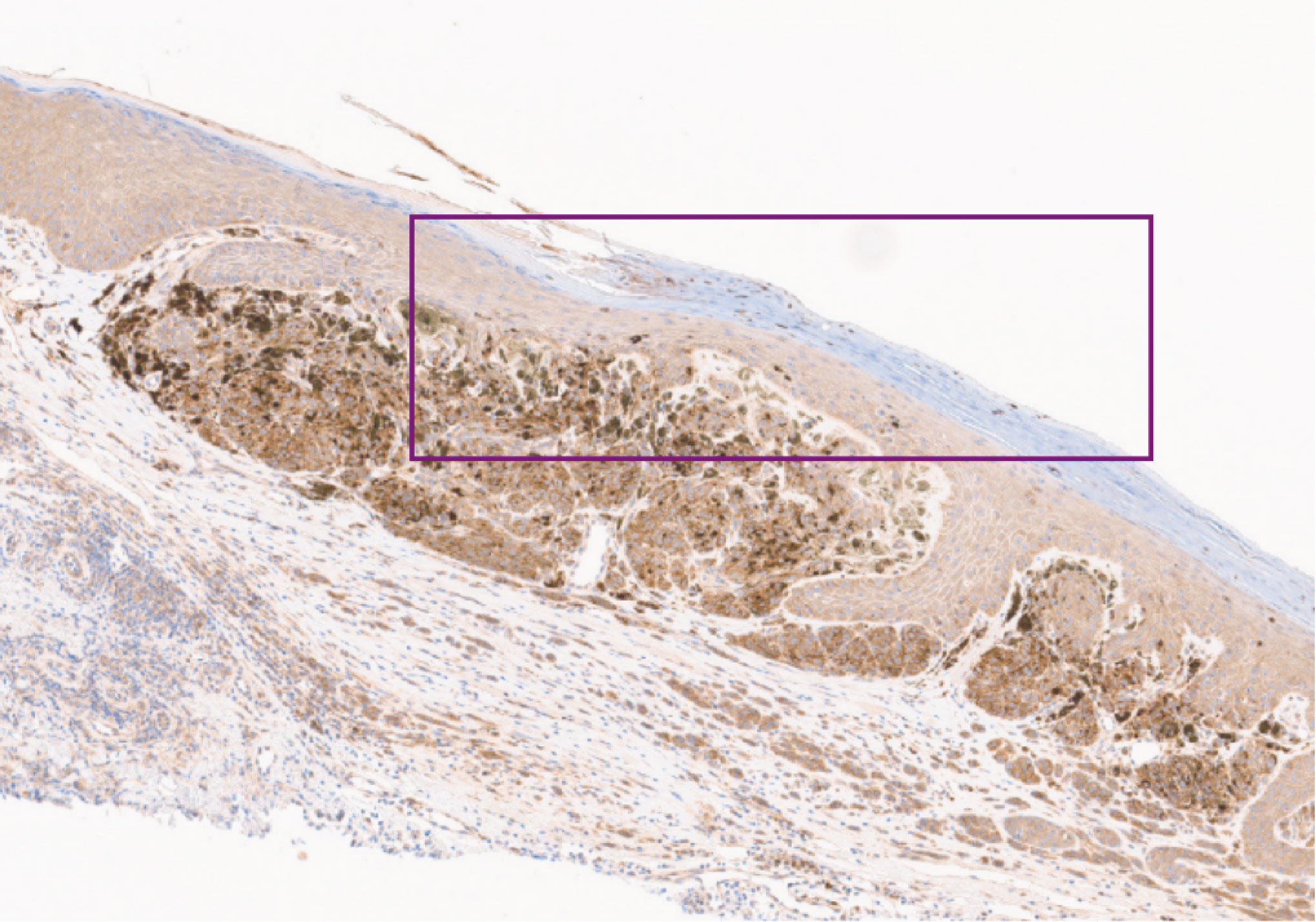
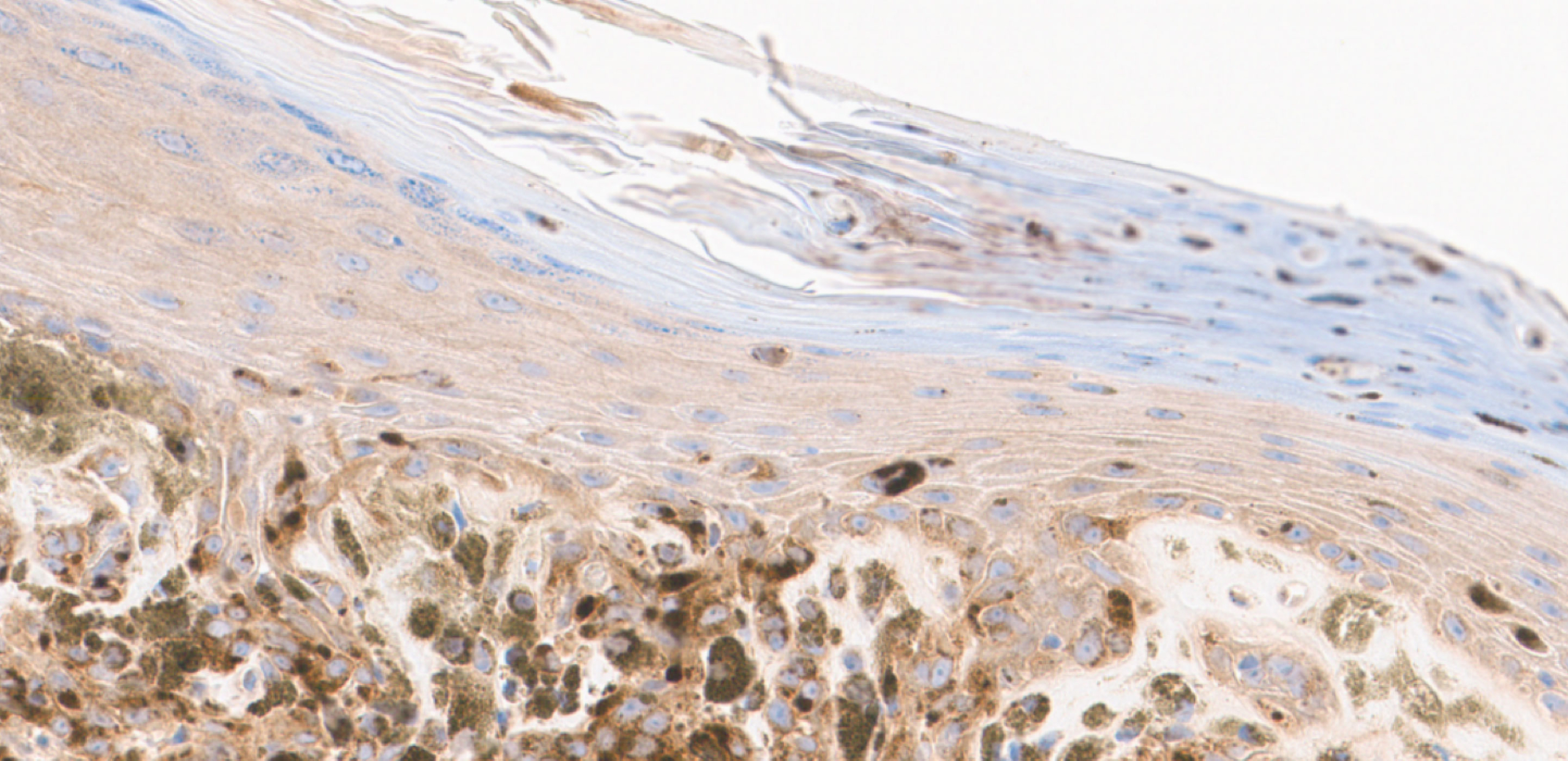
Image 2: AMBRA1 Lost x40
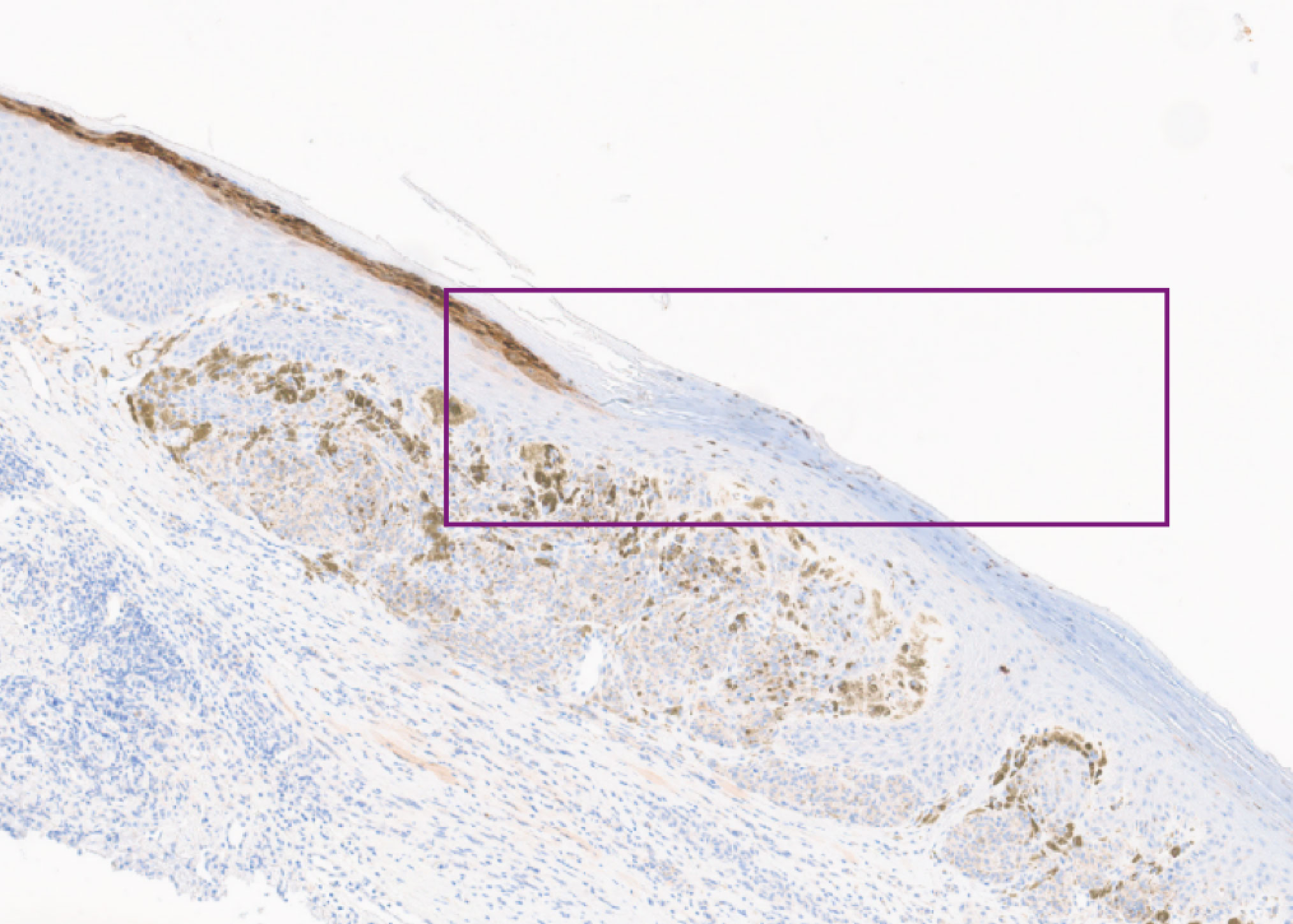
Image 3: Loricrin Lost x4

Image 4: Loricrin Lost x40
Discussion
With considerable annual spending by worldwide health economies on skin cancer care, efficiencies can be achieved by better stratification of personal risk of melanoma progression. Melanoma is the deadliest form of skin cancer, with an estimated 99,780 new cases of invasive melanoma diagnosed in the US in 2022.[1,2] Implementing AMBLor as a reflex stain (along with tyrosinase, HMB-45 and MART-1) provides a critical piece of additional information about the immediate biology of the particular lesion in question promptly following staging.
There are many factors which go into treatment plans and discussion upon diagnosis. In many cases, a sentinel lymph node biopsy (SLNB) is the next form of testing performed following initial staging. There is risk of complications when performing SLNBs including lymphoedema, seroma, hematoma, wound infection, nerve injury and deep vein thrombosis.[3] SLNB false-negatives, lesions that are recorded as benign but then reoccur, are seen in up to 21% of cases.[3] The patient in question did in fact result in one benign lymph node immunohistochemical staining on the left axillary sentinel lymph node for tyrosinase, HMB45 and MART-1 negative for metastatic melanoma. An additional screening method such as AMBLor backed by significant recurrence-free survival allows for further discussion of pursuing a more aggressive follow-up strategy and the possibility of adjuvant chemotherapy.
The latest AMBLor data, presented at ASCO 2023, revealed retention of AMBLor in melanomas (discovery cohort) which correlated with significantly increased recurrence-free survival of 96%, compared to 87% in patients for whom AMBLor was lost. [4] Similarly, the validation arm of the study confirmed retention of AMBLor was associated with increased recurrence-free survival of 98% compared to 81% for patients with stage I/II tumors in which AMBLor was lost. [4]
The patient and his family, even days before his demise, wished to continue treatment aggressively. Many discussions were held amongst the patient, his family, and his oncologist about the adjuvant therapy and how much additional disease-free survival time the patient could expect. Given that the patient continued to fight for survival following metastasis of their melanoma, initial knowledge that they had lost both AMBRA1 and Loricrin could have given the patient, his family, and treating oncologist an opportunity to revisit and further discuss the patient’s initial decision to forgo adjuvant chemotherapy.
The consistent implementation of AMBLor testing in Stage I & II non-ulcerated melanomas has many impacts for both patients and providers. Identifying low risk lesions may aid in reducing patient anxiety, positively impacting their mental health and quality of life.[5] With larger studies, a “low-risk” result may also alter follow-up strategies and ultimately may reduce the need for SLNBs subsequently lowering the risk of complications while reducing costs to the healthcare system. For patients who receive an “at-risk” result, health care providers may engage in more pointed discussions for further monitoring and adjuvant therapy options for patients. An “at-risk” test result may also warrant a closer look at a patient, with potentially lifesaving outcomes.
Finally, further study may lead to consideration for the addition of the AMBLor test to National Comprehensive Cancer Network (NCCN) Guidelines®. The test offers novel data which can support downstream decision making for patients with Stage I & II melanomas.
Disclaimer
The AMBLor test is a laboratory developed test (LDT) provided by Avero Dx and has not been reviewed by the U.S. Food & Drug Administration.
References
- American Cancer Society. Cancer Facts & Figures 2022. https://www.cancer.org/content/dam/cancer-figures.pdf Accessed June 2023.
- AIM at Melanoma. 2023 Melanoma Facts & Statistics. https://www.aimatmelanoma.org/facts-statistics/ Accessed June 2023.
- Brănisteanu DE, et al. Sentinel lymph node biopsy in cutaneous melanoma, a clinical point of view. Medicina 2022, 58, 1589. https://doi.org/10.3390/medicina58111589
- Lovat P, et al. Discovery and validation of AMBLor as a prognostic biomarker for non-ulcerated cutaneous AJCC stage I/II melanoma. J Clin Onc ASCO Abstract (Poster). 2023 Jun; 9573.
- Melanoma Patients Australia. Scanxiety. https://melanomapatients.org.au/scanxiety/ Accessed June 2023.